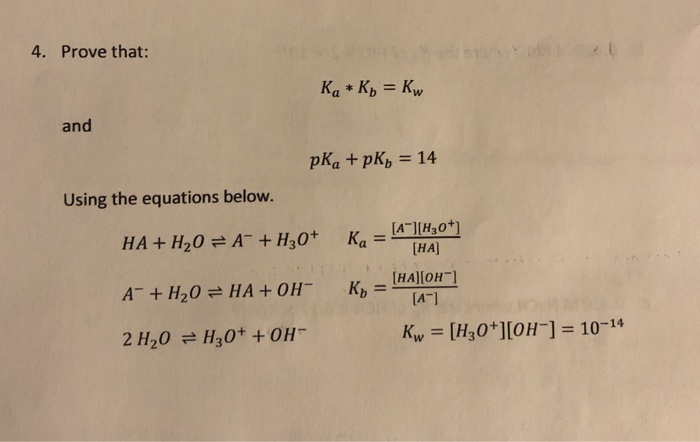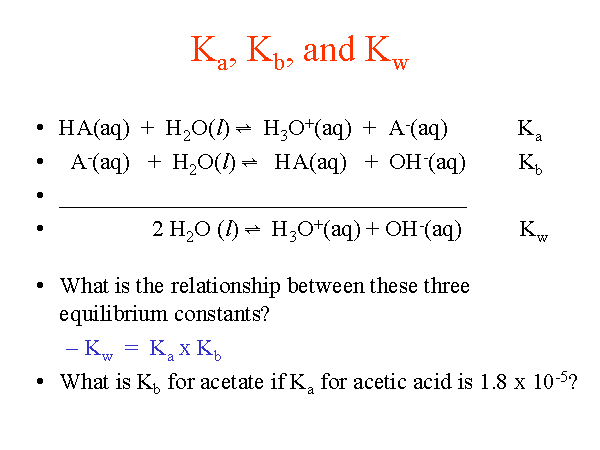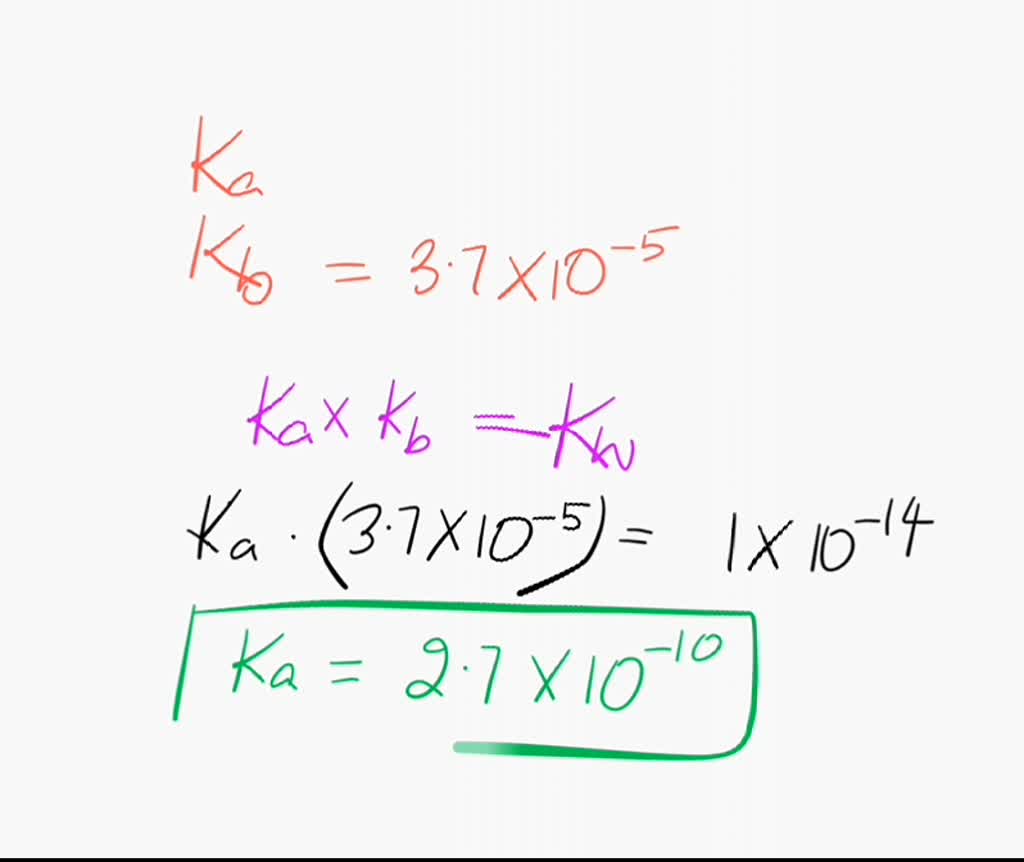
SOLVED: The Relation Between Ka and Kb of a Conjugate Acid-Base Pair A key relationship exists between the Ka Of HA and the Kb of its conjugate base; A- which we can

SOLVED: Identify the correct expression: A. Ka + Kb = Kw B. Ka - Kb = Kw C. Ka X Kb= Kw D. K = Kw Kb
What is value of Ka and kb for water (H20) and how numerically prove that ka ×kb=kw in case of water? - Quora

How is Kw=Ka x Kb if each of those equilibrium constants include the concentration of another substance in them? Kw=H3o x oh but ka which is h3o also includes another substance? :

From the dissociation constants Ka and Kb for an acid and its conjugate base, show that Ka· Kb = Kw .